Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
The Eversense® E3 Smart Transmitter, is water resistant to an Ingress Protection Rating of IP67. This means you can submerge in water 1 meter (3.2 feet) deep for up to 30 minutes. You can take showers and swim without extra protection.
Exposing the smart transmitter to conditions beyond this will result in damage and void your warranty.
The Eversense® E3 Sensor is a miniaturized fluorometer that uses fluorescent intensity to measure glucose in interstitial fluid. The sensor is implanted subcutaneously (under the skin) on the upper arm, leaving no part of the sensor protruding from the skin. The sensor remains in place and provides
CGM measurements for the operating life of the sensor for up to 180 days.
The sensor is encased in a biocompatible material and utilizes a unique fluorescent, glucose indicating polymer. A light emitting diode embedded in the sensor excites the polymer, and the polymer then rapidly signals changes in glucose concentration via a change in light output. The measurement is then relayed to the smart transmitter. Measurements are completed automatically and require no action by the user.
The sensor is approximately 3.5 mm x 18.3 mm and has a silicone ring that contains a small amount of dexamethasone acetate, an anti-inflammatory steroid drug. The dexamethasone acetate minimizes inflammatory responses, very similar to common medical devices, such as pacemakers.
The Eversense E3 Sensor includes the sacrificial boronic acid (SBA) design modification.

There may be times when you wish to temporarily stop sharing data with a member, but not remove them from your Circle.
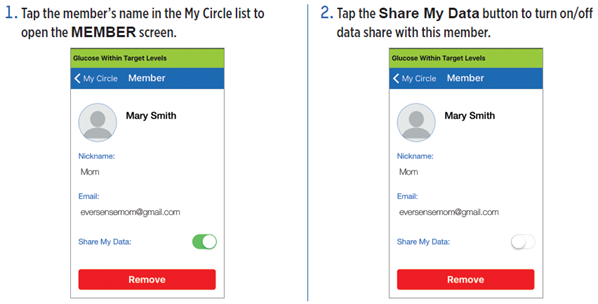
IMPORTANT: If you have disabled the Share My Data feature for a member, that member will not see any of your Eversense® CGM System glucose data, alerts or event history. Members will see your status as Offline on their Eversense® NOW App when you have disabled the Share my Data feature. It can take up to 10 minutes for the change to display on the Eversense® NOW user’s app.
Your Eversense® E3 CGM System will alert you when your glucose levels are outside the alert settings you choose. When you have reached your low and high glucose alert levels, the smart transmitter vibrates, and the mobile app gives an audible alert as well as displays a message on the screen. You should immediately perform a finger stick blood glucose test before making a treatment decision.

To unpair an old transmitter, go to the Main Menu in the App and tap Connect. Tap and hold the
name of your old smart transmitter. Tap OK to stop the app from automatically connecting with the old smart transmitter. Follow the steps in the user guide for linking the new transmitter to your sensor.
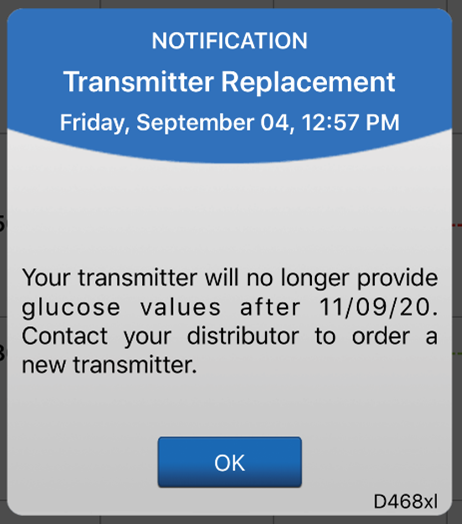
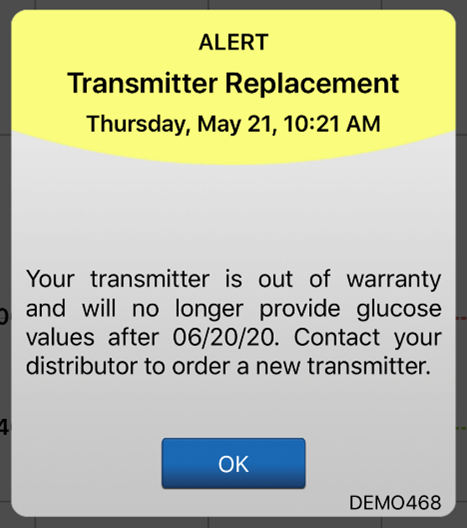
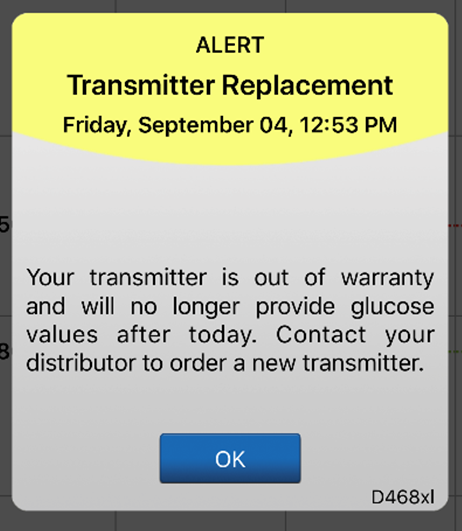
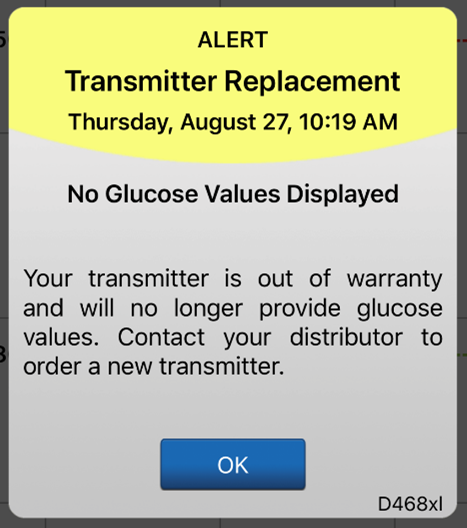
The system will send out a notification on day 330, and alerts on days 366, 395, and 396 reminding the user to order a new smart transmitter. After day 395, the smart transmitter will no longer be able to provide CGM data to you.
Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
MKT-001692 Rev 1