Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
The Bluetooth® communication distance between app and smart transmitter is up to 25 feet (7.6 meters).
Wireless communication to the app will not function well when communicating through water. The range will decrease if you are on a waterbed, in a bath, pool, etc. The communication range may also be reduced in certain environments, especially those with multiple electronic devices nearby.
This should only be done if directly instructed by customer care. The system will return to initialization phase after re-linking is complete.
Speak to your health care professional to determine your personal high and low glucose alert levels. Do not change your glucose alert levels without consulting with your health care professional.
The high and low glucose levels in the app are pre-set, but can be customized to your preferences. Based on the preset (default) glucose levels:
“HI” Reading Indicates a sensor glucose reading is greater than 400 mg/dL
“LO” Reading Indicates sensor glucose reading is less than 40 mg/dL
Operational Conditions: 5 - 40 °C (41 - 104 °F)
Storage Conditions: 0 - 35 °C (32 - 95 °F)
Cabin Pressure: 700hPa to 1060hPa
Altitude: 10,000 feet (3,048 meters)
Relative Humidity Range (non-condensing): 15% to 90%
Protection Against Electrical Shock: Type BF applied part
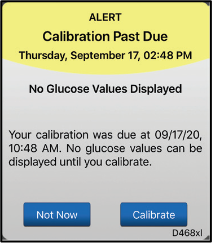
If you do not have access to a blood glucose meter when you receive a calibration prompt, your Eversense E3 CGM system will continue to provide glucose readings for up to 4 additional hours. You should take action during this time to obtain your blood glucose meter. After the 4 hours, you will receive a Calibration Past Due alert and no glucose values will be displayed until the next calibration is entered.
Even with a non-adjunctive CGM system, you should always carry a blood glucose meter with you as you should always be prepared to take a blood glucose test when prompted for a calibration or when any of the following situations occur:
- Your symptoms do not match the glucose value displayed on your app
- No glucose value is displayed (during system initialization, for example)
- Your system prompts you to use your BG meter for making a treatment decision in the app status bar
- You are taking medications of the tetracycline class
Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
MKT-001692 Rev 1