Your Questions, Answered
We’ve assembled the most frequently asked questions about Eversense into one place. Got a question that you can’t find the answer to? Contact us.
Eversense® NOW is a mobile App that allows the Eversense® users to share their data and information with friends and family. The Eversense® NOW user will be able to monitor the CGM glucose data of up to 10 Eversense® CGM users.
Note: The Eversense® CGM user has the ability to add/remove friends and family from Eversense NOW access to their data at any time.
The Charge Transmitter alert appears once the battery reaches 10% power remaining. Once the alert appears, the smart transmitter has approximately 2 hours of power left.
It is important to charge your battery when you receive the “Charge Transmitter” alert.
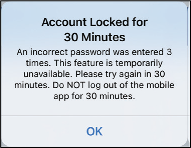
If you enter an incorrect password three times on your DMS log in page, some mobile app features may be unavailable for 30 minutes, including no data syncing to your DMS account, no Eversense NOW users can be added to your Circle, and no changes to your profile picture can be made. You should not log out of the mobile app during this time.
The sensor incision site usually heals within a few days.
Always follow your health care professional’s instructions for care after the sensor insertion or removal.
Contact your health care professional if you have pain, redness, or swelling at the incision site(s) later than 5 days after the sensor insertion or removal.
If you are concerned about incision site reactions at any time, contact your health care professional.
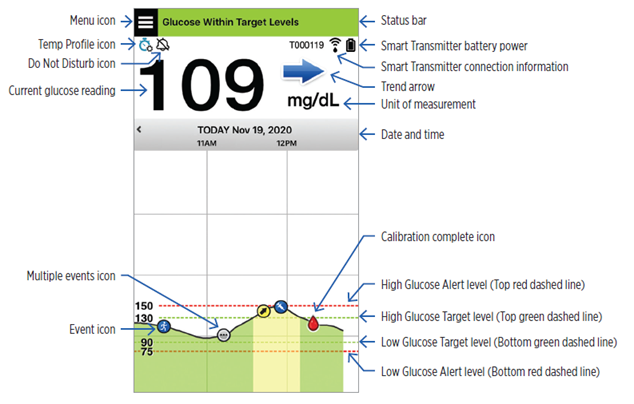
The arrows on the screen are called “Trend Arrows”. The trend arrow shows the current direction the glucose is going and how fast the glucose trend is changing.
The app uses the last 20 minutes of continuous glucose data for calculating glucose trends. When there are not enough sensor values available for the calculation, the arrow will display as grey.

Still have questions?
The Eversense team is dedicated and ready to provide the answers and support you need. Simply click the link below.
The Eversense® E3 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to 180 days in persons with diabetes age 18 and older. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are still required for calibration primarily one time a day after day 21, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense E3 CGM System is a prescription device; patients should talk to their health care provider to learn more.
The Eversense® 365 Continuous Glucose Monitoring (CGM) System is indicated for continually measuring glucose levels for up to one year in people (18 years and older) with diabetes. The system is indicated for use to replace fingerstick blood glucose (BG) measurements for diabetes treatment decisions. Fingerstick BG measurements are required for calibration one time a week after day 13, and when symptoms do not match CGM information or when taking medications of the tetracycline class. The sensor insertion and removal procedures are performed by a health care provider. The Eversense 365 CGM System is a prescription device; patients should talk to their health care provider to learn more.
For important safety information, see bit.ly/eversensesafety
Eversense, Eversense E3 Continuous Glucose Monitoring, Eversense 365 Continuous Glucose Monitoring, and the Eversense logo are trademarks of Senseonics, Incorporated. All other trademarks are properties of their respective owners and are used solely for informative purposes. No relationship or endorsement should be inferred or implied.
Apple Watch® is a product of Apple, Inc., and may be separately purchased from an authorized Apple retailer. Apple Watch is not included with the Eversense CGM System. Android is a trademark of Google LLC.
MKT-001692 Rev 1